 
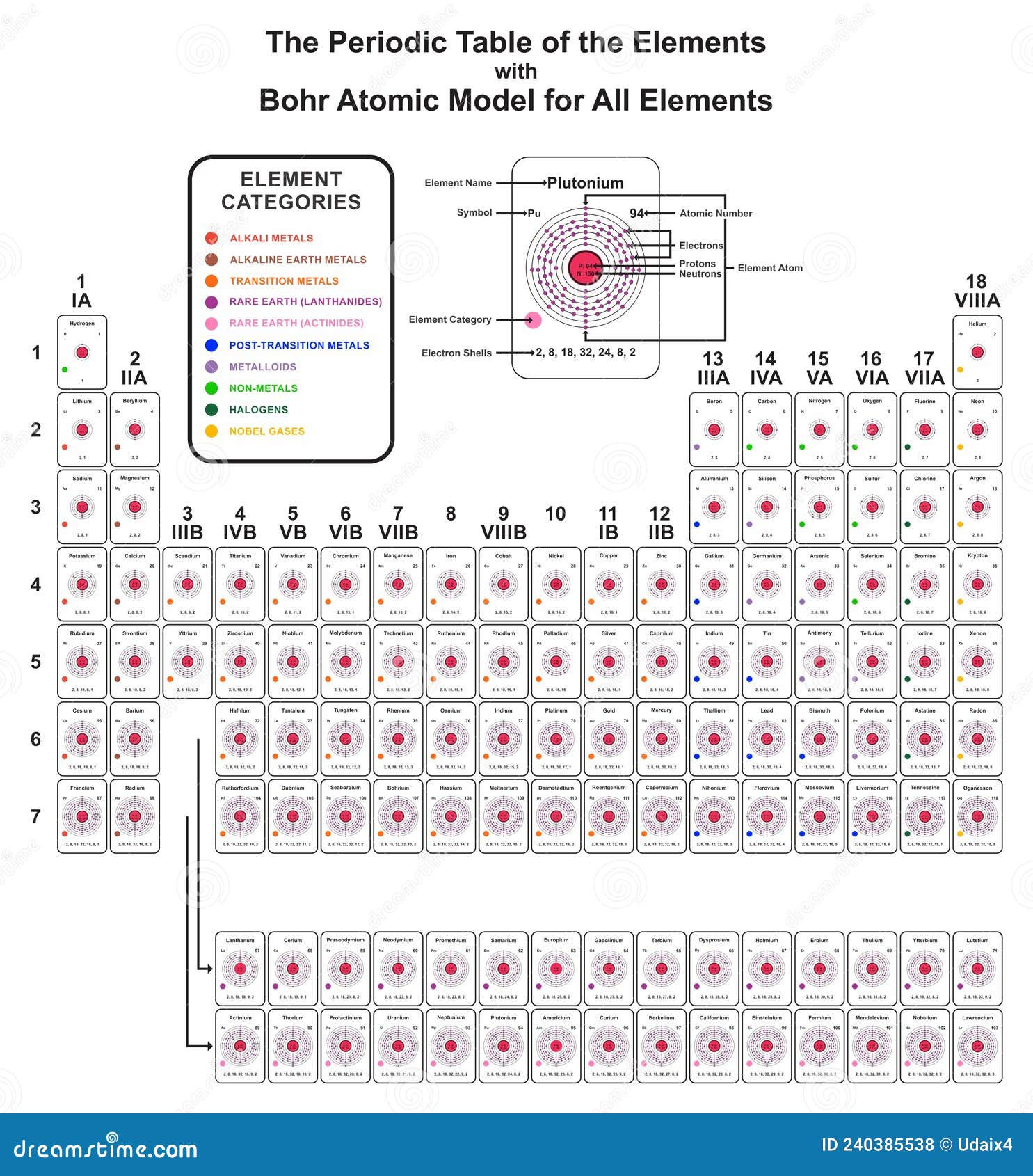
Now, put the values in the above-mentioned formula: Number of neutrons = Atomic mass (rounding it up to the nearest whole number) – Number of protonsĪs mentioned in the chlorine box drawn above the atomic mass of the chlorine atom is 35.353.Īfter rounding it up to the nearest whole number we get 35. Now, we will calculate the number of neutrons present in the chlorine atom.įor any atom number of neutrons present in the nucleus is given by the following formula: Therefore, for the chlorine atom, the number of protons = atomic number = 17 In the case of the chlorine atom, the atomic number is 17. The number of protons for any atom is equal to the atomic number of that atom. Let us first start with determining the number of protons in chlorine. To draw the Bohr model of chlorine we will first have to find out the atomic species contained in this atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
